Targeting the cancer cells that resist other therapies
Solid tumors develop zones that lack oxygen. Normoxic cancer cells have normal oxygen levels and respond to treatment. Hypoxic cancer cells survive in oxygen-starved environments — and resist radio-, chemo-, and immunotherapy. When these cells survive, they regrow the tumor. This is why patients relapse.
PH1154 - A first-in-class hypoxia-targeting macrocycle
PH1154 is a first-in-class drug candidate that selectively targets low-oxygen cancer cells by disrupting their energy production, tackling a fundamental cause of treatment failure.

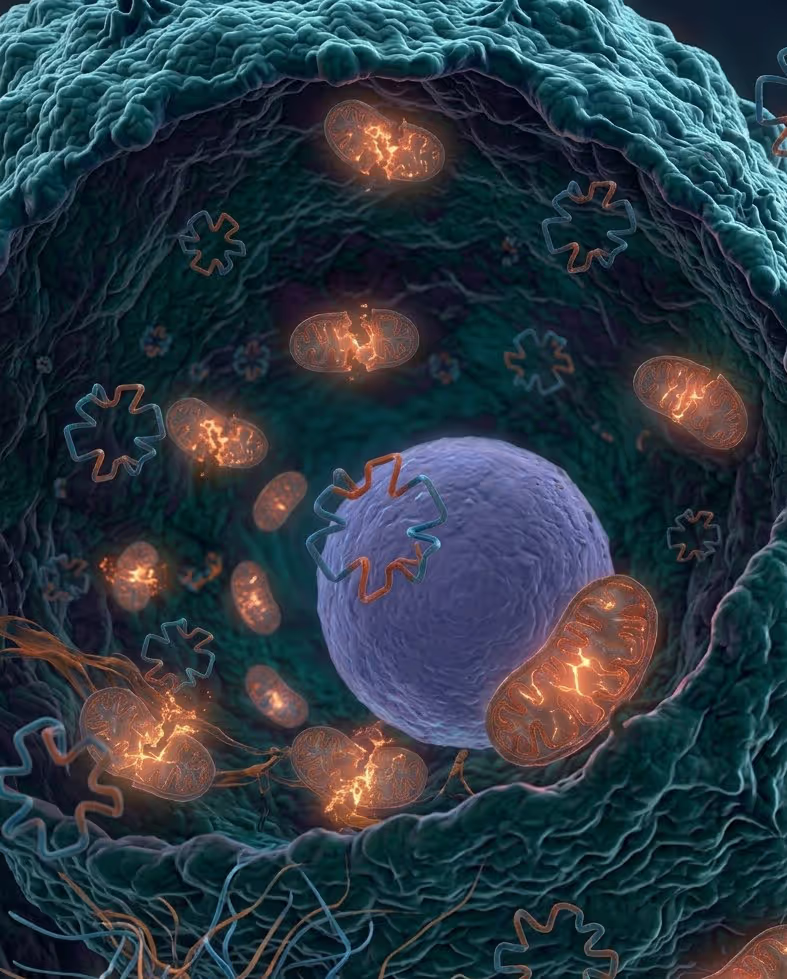
Why hypoxic cancer cells resist treatment
Hypoxic cancer cells have adapted to survive in low-oxygen conditions — and this adaptation makes them resistant to standard treatment approach.
More than 3 million patients receive radiotherapy each year. For many, hypoxic cancer cells are the reason treatment fails.
Radiotherapy
Relies on oxygen to damage DNA. In hypoxic areas, the radiation is inefficient.
Chemotherapy
Targets rapidly dividing cells. But hypoxic cells slow their metabolism and become dormant — insensitive to chemotherapy.
Immunotherapy
Struggles to penetrate the immunosuppressive environment that hypoxic cells create around themselves.
The consequence
Hypoxic cancer cells survive treatment, remain dormant, and eventually regrow — driving tumor relapse and metastasis.
PH1154
A new approach to an old problem
PH1154 is a first-in-class hypoxia-targeting macrocycle (HTMC) designed to do what conventional treatments cannot: selectively eliminate hypoxic cancer cells.
How it works
Hypoxic cancer cells rewire their metabolism to survive without oxygen. This creates abnormally high electron levels in their mitochondria — a vulnerability unique to these cells.
PH1154 exploits this. It triggers the release of toxic radicals that destroy the cell from within. In oxygen-rich environments, PH1154 remains inactive and has no effect on healthy tissue.
Built for purpose
PH1154 is inspired from BE-43547, a natural compound with hypoxia-selective properties. Through fundamental redesign, we created a molecule optimized for hypoxia-selectivity, safety, IP positioning, and production cost.
Hypoxia-targeting macrocycles (HTMCs) are first-in-class small molecules that are designed to exploit all the DMPK benefits that comes with macrocyclic drug candidates, such as optimal potency, enhanced circulation and tissue penetration as well as reduced metabolic degradation.
What makes it different
Previously, attempts to target tumor hypoxia relied primarily on hypoxia-activated prodrugs (HAPs). HAPs are designed to release DNA-damaging chemotherapy in hypoxic tumor tissue - a virtually chemotherapy-resistant part of tumors.
PH1154 works differently by disrupting mitochondrial adaptions to hypoxia. This works through a biochemically induced mechanism with no protein target. This makes it extremely difficult to develop resistance to PH1154.


Strong preclinical results
PH1154 has demonstrated potent anti-tumor activity in preclinical in vivo and in vitro models — with particularly striking results in combination with radiotherapy.
Combination with radiotherapy
In a mouse model, PH1154 combined with radiotherapy achieved 50% tumor-free survival. Radiation alone achieved just 1 in 8.
Broad activity across cancer types
PH1154 was tested against 35 cancer cell lines spanning diverse tissues, sexes, and ancestries. 89% responded with hypoxia-selective toxicity, confirming activity across a wide range of solid tumors.
Key developments
Kripthonite Therapeutics is in preclinical development, advancing PH1154 toward clinical trials.
We have established preclinical proof-of-concept for PH1154 as a hypoxia-targeted therapy. Current work focuses on efficacy studies, safety evaluation, and preparation for first-in-human studies.
First-in-class molecule series discovered and patented
Successful hit to lead development
Validated activity in spheroid and 2D in vitro models
Unprecedented hypoxia-selectivity demonstrated
Compelling in vivo PoC established

Connect with us
We are seeking partnerships with strong players in oncology who share our mission to bring truly curative treatments to patients with solid tumors.
